"Smart" Molecular Self-Assembly for Safer, Longer-Lasting Solid-State Batteries
FAYETTEVILLE, GA, UNITED STATES, March 3, 2026 /EINPresswire.com/ -- Researchers from Tsinghua University explore how supramolecular self-assembly can revolutionize solid-state lithium batteries. By utilizing reversible non-covalent interactions like hydrogen bonding, they have developed dynamic electrolyte networks with self-healing capabilities and high ionic conductivity. This framework effectively suppresses lithium dendrite growth and reduces interfacial impedance, offering a transformative strategy for next-generation, high-safety energy storage systems for electric vehicles.
As the global demand for electric vehicles and portable electronics surges, high-energy-density and inherently safe energy storage systems has become more important than ever. However, while solid-state lithium batteries (SSLBs) offer high safety due to their non-flammability, traditional solid electrolytes face significant bottlenecks, including low ionic conductivity, poor interfacial contact, and mechanical brittleness.
In a review published in Supramolecular Materials, a team of researchers from China highlight a new approach: using supramolecular chemistry to engineer "smart" battery components. The study provides a molecular engineering foundation for realizing practical, high-efficiency, and safe next-generation batteries.
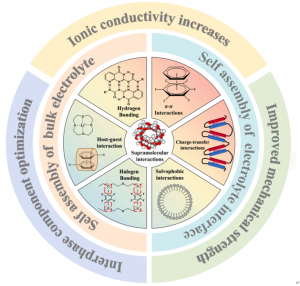
“Unlike traditional materials that rely on rigid covalent bonds, supramolecular materials utilize reversible non-covalent interactions such as hydrogen bonding, halogen bonding, and π-π stacking to create highly ordered, self-assembled structures,” explains senior and corresponding author Kai Liu.
Notably, supramolecular chemistry provides a programmable molecular-level design framework for solid-state batteries. “These dynamic interactions act as a ‘smart glue,’ allowing electrolytes to self-heal microcracks and adapt to the volume changes of electrodes during cycling,” adds Liu. “This flexibility is crucial for suppressing lithium dendrite growth, which often leads to short circuits in conventional designs."
The researchers also detailed how these molecular interactions build efficient ion transport pathways, lowering energy barriers and improving the battery's rate performance. “By precisely regulating the interfacial composition, supramolecular strategies significantly reduce impedance and enhance long-term cycling stability,” says Liu.
References
DOI
10.1016/j.supmat.2025.100118
Original Source URL
https://doi.org/10.1016/j.supmat.2025.100118
Funding Information
This research was supported by the Tsinghua University-China Petrochemical Corporation Joint Institute for Green Chemical Engineering (224247) and the Tsinghua-Toyota Joint Research Fund.
Lucy Wang
BioDesign Research
email us here
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.